INDICATION: QUZYTTIR is indicated for the treatment of acute
urticaria in adults and children 6 months of age and older.
Adverse reactions for QUZYTTIR and IV DPH injection in the phase 3 study1,2
The most common adverse reactions (incidence less than 1%) with QUZYTTIR include dysgeusia, headache,
paresthesia, presyncope, dyspepsia, feeling hot, and hyperhidrosis1

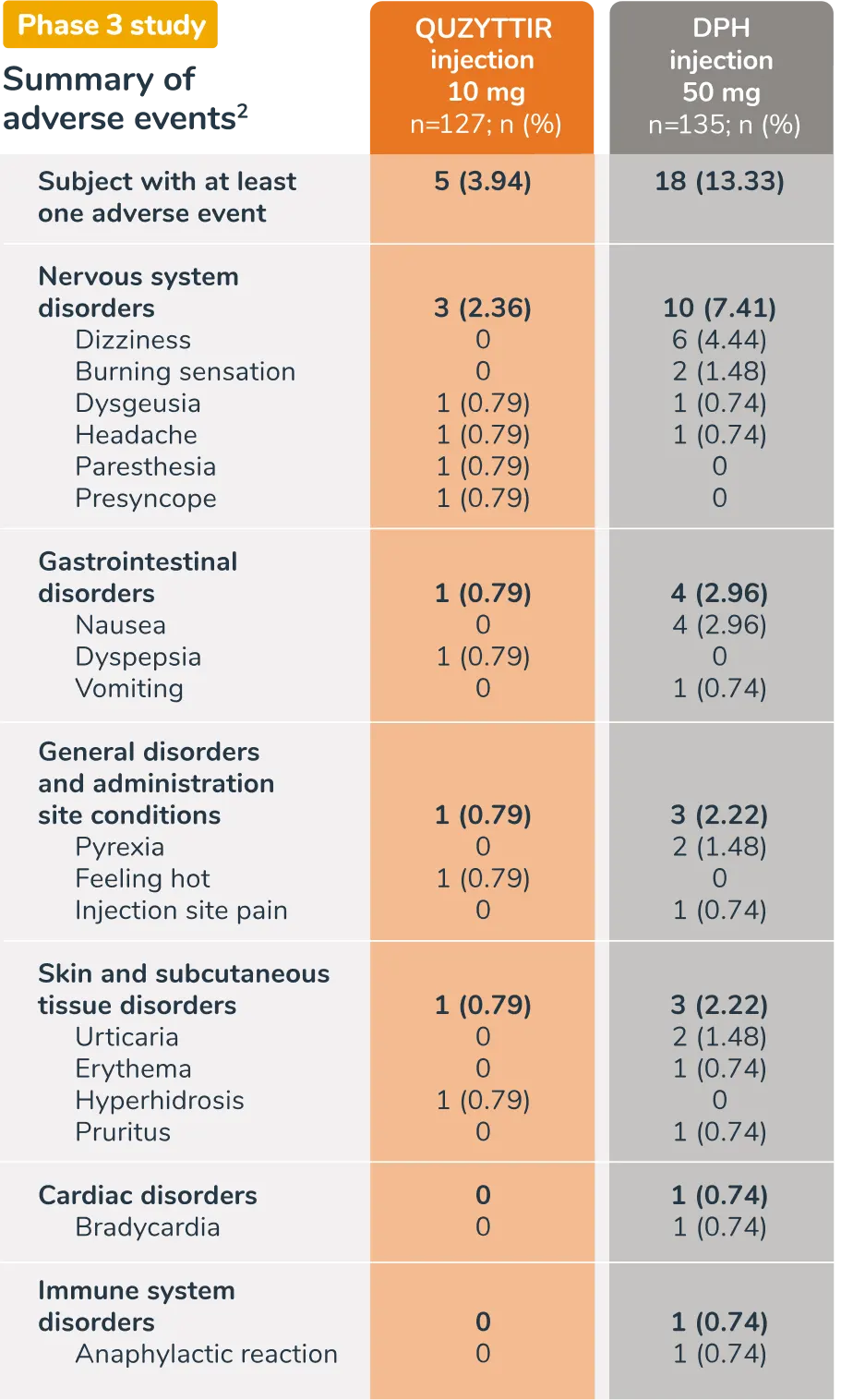
| Adverse Event | QUZYTTIR injection 10mg (n=127) | DPH injection 50 mg (n=135) |
|---|---|---|
| Subject with at least one adverse event | 5 (3.94) | 18 (13.33) |
| Nervous system disorders | 2 (2.36) | 10 (7.41) |
| Dizziness | 0 | 6 (4.44) |
| Burning sensation | 0 | 2 (1.48) |
| Dysgeusia | 1 (0.79) | 1 (0.74) |
| Headache | 1 (0.79) | 1 (0.74) |
| Paresthesia | 1 (0.79) | 0 |
| Presyncope | 1 (0.79) | 0 |
| Gastrointestinal disorders | 1 (0.79) | 4 (2.96) |
| Nausea | 1 (0.79) | 4 (2.96) |
| Dyspepsia | 0 | 0 |
| Vomiting | 1 (0.79) | 1 (0.74) |
| General disorders and administration site conditions | 1 (0.79) | 3 (2.22) |
| Pyrexia | 0 | 2 (1.48) |
| Feeling hot | 1 (0.79) | 0 |
| Injection site pain | 0 | 1 (0.74) |
| Skin and subcutaneous tissue disorders | 1 (0.79) | 3 (2.22) |
| Urticaria | 0 | 2 (1.48) |
| Erythema | 0 | 1 (0.74) |
| Hyperhidrosis | 1 (0.79) | 0 |
| Pruritus | 0 | 1 (0.74) |
| Cardiac disorders | 0 | 1 (0.74) |
| Bradycardia | 0 | 1 (0.74) |
| Immune system disorders | 0 | 1 (0.74) |
| Anaphylactic reaction | 0 | 1 (0.74) |

